Detection of Circulating Tumor Cells in Patients With Adrenocortical Carcinoma: A Monocentric Preliminary Study
Context:
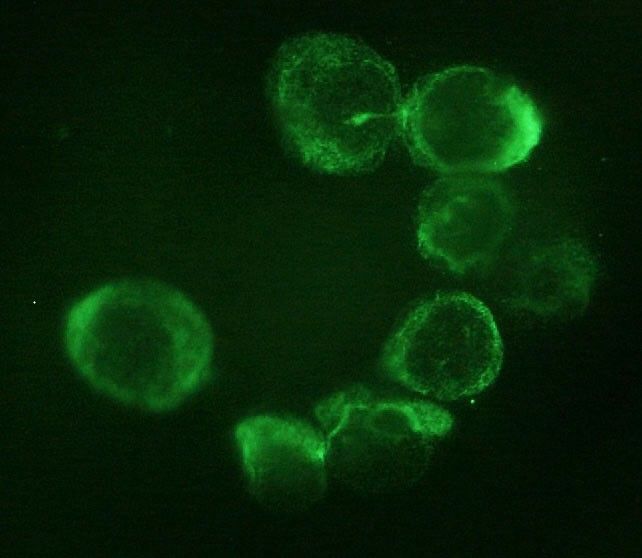
Adrenocortical carcinoma (ACC) is a rare malignancy, the prognosis of which is mainly dependent on stage at diagnosis. The identification of disease-associated markers for early diagnosis and drug monitoring is mandatory. Circulating tumor cells (CTCs) are released into the bloodstream from primary tumor/metastasis. CTC detection in blood samples may have enormous potential for assisting in the diagnosis of malignancy, estimating prognosis, and monitoring the disease.
Objective:
The aim of the study was to investigate the presence of CTCs in blood samples of patients with ACC or benign adrenocortical adenoma (ACA).
Setting:
We conducted the study at a university hospital.
Intervention:
CTC analysis was performed in blood samples from 14 ACC patients and 10 ACA patients. CTCs were isolated on the basis of cell size by filtration through ScreenCell devices, followed by identification according to validated morphometric criteria and immunocytochemistry.
Main Outcome Measure:
We measured the difference in CTC detection between ACC and ACA.
Results:
CTCs were detected in all ACC samples, but not in ACA samples. Immunocytochemistry confirmed the adrenocortical origin. When ACC patients were stratified according to the median value of tumor diameter and metastatic condition, a statistically significant difference was found in the number of CTCs detected after surgery. A significant correlation between the number of CTCs in postsurgical samples and clinical parameters was found for tumor diameter alone.
Conclusions:
Our findings provide the first evidence for adrenocortical tumors that CTCs may represent a useful marker to support differential diagnosis between ACC and ACA. The correlation with some clinical parameters suggests a possible relevance of CTC analysis for prognosis and noninvasive monitoring of disease progression and drug response.
Thoracic surgery in the UK

No abstract available
A new device for rapid isolation by size and characterization of rare circulating tumor cells

Background: Circulating tumor cells (CTCs) likely derive from clones in the primary tumor, suggesting that they can be used for all biological tests applying to the primary cells. Materials and Methods: The ScreenCell® devices are single-use and low-cost innovative devices that use a filter to isolate and sort tumor cells by size. Results: The ScreenCell® Cyto device is able to isolate rare, fixed, tumor cells, with a high recovery rate. Cells are well preserved morphologically. Immunocytochemistry and FISH assays can be performed directly on the filter. The ScreenCell® CC device allows isolation of live cells able to grow in culture. High quality genetic materials can be obtained directly from tumor cells isolated on the ScreenCell® MB device filter. Conclusion: Due to their reduced size, versatility, and capacity to isolate CTCs within minutes, the ScreenCell® devices may be able to simplify and improve non-invasive access to tumor cells.
Rapid and Simple Isolation of Circulating Tumor Cells for Clinical and Research Applications Using ScreenCell

Circulating tumor cells (CTCs) are malignant cells shed by the primary tumor or metastases into the peripheral circulation. CTCs give rise to distant metastases that are usually the ultimate cause of cancer-related death. A significant proportion of patients with early stage cancer in whom no metastases are identifiable will ultimately relapse as a result of hematogenous spread of tumor cells that were undetected at initial diagnosis and treatment.
Rapid separation of mononuclear hodgkin from multinuclear reed-sternberg cells

We describe a method to isolate small mononucleated Hodgkin (H) cells from multinucleated Reed Sternberg (RS) cells of Hodgkin lymphoma using the ScreenCell filter device. This filtration-based approach lends itself to future clinical applications in that it enables the separation of H and RS cells from lymph node biopsies, bone marrow aspirates, pleural effusions, and blood, including the isolation of monoclonal Hodgkin precursor cells from the blood.
Circulating tumour cell enumeration does not correlate with Miller-Payne grade in a cohort of breast cancer patients undergoing neoadjuvant chemotherapy
Purpose: The association between pathological complete response (pCR) in patients receiving neoadjuvant chemotherapy (NAC) for breast cancer and Circulating Tumour Cells (CTCs) is not clear. The aim of this study was to assess whether CTC enumeration could be used to predict pathological response to NAC in breast cancer as measured by the Miller-Payne grading system.
Methods: Twenty-six patients were recruited, and blood samples were taken pre- and post-NAC. CTCs were isolated using the ScreenCell device and stained using a modified Giemsa stain. CTCs were enumerated by 2 pathologists and classified as single CTCs, doublets, clusters/microemboli and correlated with the pathological response as measured by the Miller-Payne grading system. χ2 or ANOVA was performed in SPSS 24.0 statistics software for associations.
Results: 89% of patients had invasive ductal carcinoma (IDC) and 11% invasive lobular carcinoma (ILC). At baseline 85% of patients had CTCs present, median 7 (0-161) CTCs per 3 ml of whole blood. Post-chemotherapy, 58% had an increase in CTCs. This did not correlate with the Miller-Payne grade of response. No significant association was identified between the number of CTCs and clinical characteristics; however, we did observe a correlation between pre-treatment CTC counts and body mass index, p < 0.05. Conclusions: Patients with a complete response to NAC still had CTCs present, suggesting enumeration is not sufficient to aid surgery stratification. Additional characterisation and larger studies are needed to further characterise CTCs isolated pre- and post-chemotherapy. Long-term follow-up of these patients will determine the significance of CTCs in NAC breast cancer patients.
Advancing Risk Assessment of Intermediate Risk Prostate Cancer Patients
The individual risk to progression is unclear for intermediate risk prostate cancer patients. To assess their risk to progression, we examined the level of genomic instability in circulating tumor cells (CTCs) using quantitative three-dimensional (3D) telomere analysis. Data of CTCs from 65 treatment-naïve patients with biopsy-confirmed D’Amico-defined intermediate risk prostate cancer were compared to radical prostatectomy pathology results, which provided a clinical endpoint to the study and confirmed pre-operative pathology or demonstrated upgrading. Hierarchical centroid cluster analysis of 3D pre-operative CTC telomere profiling placed the patients into three subgroups with different potential risk of aggressive disease. Logistic regression modeling of the risk of progression estimated odds ratios with 95% confidence interval (CI) and separated patients into “stable” vs. “risk of aggressive” disease. The receiver operating characteristic (ROC) curve showed an area under the curve (AUC) of 0.77, while prostate specific antigen (PSA) (AUC of 0.59) and Gleason 3 + 4 = 7 vs. 4 + 3 = 7 (p > 0.6) were unable to predict progressive or stable disease. The data suggest that quantitative 3D telomere profiling of CTCs may be a potential tool for assessing a patient’s prostate cancer pre-treatment risk.
Strategies for Isolating and Propagating Circulating Tumor Cells in Men with Metastatic Prostate Cancer

Selecting a well-suited method for isolating/characterizing circulating tumor cells (CTCs) is challenging. Evaluating sensitive and specific markers for prostate cancer (PCa)-specific CTC identification and analysis is crucial. We used the CellCollector EpCAM-functionalized system (CC-EpCAM) and evaluated and developed a PCa-functionalized version (CC-PCa); we then compared CTC isolation techniques that exploit the physical and biological properties of CTCs. We established two cohorts of metastatic PCa patients (mPCa; 15 in cohort 1 and 10 in cohort 2). CTC cultivation experiments were conducted with two capturing methods (Ficoll and ScreenCell). The most sensitive detection rates and highest CTC counts were reached with the CC-PCa and ScreenCell system. Patients with ≥5 CTCs isolated with CC-EpCAM had an overall survival (OS) of 0.93 years, and patients with ≥5 CTCs isolated with CC-PCa had an OS of 1.5 years in cohort 1. Nevertheless, we observed the highest sensitivity and specificity for 24-month survival by the Ficoll with CD45 depletion and ScreenCell system with May-Grunwald Giemsa (MGG) staining. The EpCAM molecule is an essential factor related to OS for CTC isolation based on biological properties in mPCa patients. The best-suited CTC capture system is not limited to one characteristic of cells but adapted to downstream analysis.
Detection of Circulating Tumour Cells in Urothelial Cancers and Clinical Correlations: Comparison of Two Methods
Circulating tumour cells (CTC) are identified exploiting their protein/gene expression patterns or distinct size compared to blood cells. Data on CTC in bladder cancer (BC) are still scarce. We comparatively analyzed CTC enrichment by AdnaTest ProstateCancerSelect (AT) and ScreenCell®Cyto (SC) kits, combined with identification by EPCAM, MUC1, and ERBB2 expression and by cytological criteria, respectively, in 19 nonmetastatic (M0) and 47 metastatic (M+) BC patients, at baseline (T0) and during treatment (T1). At T0, CTC positivity rates by AT were higher in M+ compared to M0 cases (57.4% versus 25%, p = 0.041). EPCAM was detected in 75% of CTC-positive samples by AT, showing increasing expression levels from T0 to T1 (median (interquartile range, IQR): 0.18 (0.07-0.42) versus 0.84 (0.33-1.84), p = 0.005) in M+ cases. Overall, CTC positivity by SC was around 80% regardless of clinical setting and time point of analysis, except for a lower occurrence at T1 in M0 cases. At T0, circulating tumour microemboli were more frequently (25% versus 8%) detected and more numerous in M+ compared to M0 patients. The approach used for CTC detection impacts the outcome of CTC studies. Further investigations are required to clarify the clinical validity of AT and SC in specific BC clinical contexts.
Liquid Biopsy in Rare Cancers: Lessons from Hemangiopericytoma

Hemangiopericytoma (HPT) is a rare mesenchymal tumor of fibroblastic type and for its rarity is poorly studied. The most common sites of metastatic disease in patients with intracranial HPT are the bone, liver, and lung, suggestive for an hematogenous dissemination; for this reason, we investigated, for the first time, the presence of circulating tumor cells (CTCs) in hemangiopericytoma patient by CellSearch® and SceenCell® devices. Peripheral blood samples were drawn and processed by CellSearch, an EpCAM-dependent device, and ScreenCell®, a device size based. We found nontypical CTCs by CellSearch system and the immunofluorescence analysis performed on CTCs isolate by ScreenCell demonstrated the presence of single CTCs and CTC clusters. The molecular characterization of single CTCs and CTC clusters, using antibodies directed against EpCAM, CD34, cytokeratins (8, 18, and 19), and CD45, showed a great heterogeneity in CTC clusters. We believe that the present study may open a new scenario in the rare tumors: the introduction of the liquid biopsy and the molecular characterization of circulating tumor cells could lead to personalized targeted treatments and also for rare tumors.