Detection of Circulating Tumor Cells in Patients With Adrenocortical Carcinoma: A Monocentric Preliminary Study
Context:
Adrenocortical carcinoma (ACC) is a rare malignancy, the prognosis of which is mainly dependent on stage at diagnosis. The identification of disease-associated markers for early diagnosis and drug monitoring is mandatory. Circulating tumor cells (CTCs) are released into the bloodstream from primary tumor/metastasis. CTC detection in blood samples may have enormous potential for assisting in the diagnosis of malignancy, estimating prognosis, and monitoring the disease.
Objective:
The aim of the study was to investigate the presence of CTCs in blood samples of patients with ACC or benign adrenocortical adenoma (ACA).
Setting:
We conducted the study at a university hospital.
Intervention:
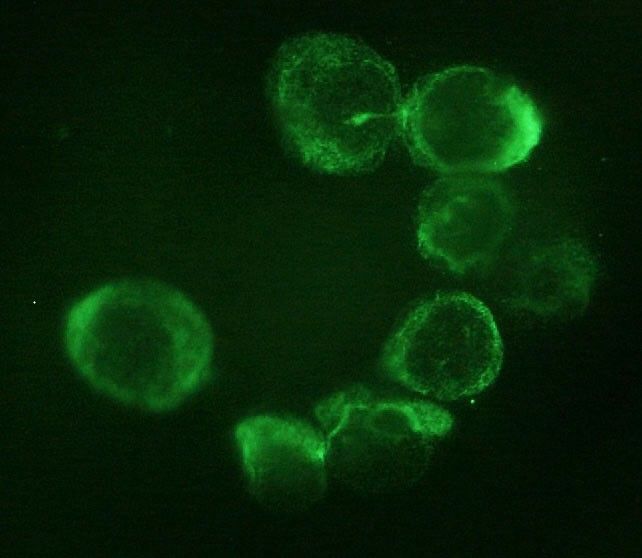
CTC analysis was performed in blood samples from 14 ACC patients and 10 ACA patients. CTCs were isolated on the basis of cell size by filtration through ScreenCell devices, followed by identification according to validated morphometric criteria and immunocytochemistry.
Main Outcome Measure:
We measured the difference in CTC detection between ACC and ACA.
Results:
CTCs were detected in all ACC samples, but not in ACA samples. Immunocytochemistry confirmed the adrenocortical origin. When ACC patients were stratified according to the median value of tumor diameter and metastatic condition, a statistically significant difference was found in the number of CTCs detected after surgery. A significant correlation between the number of CTCs in postsurgical samples and clinical parameters was found for tumor diameter alone.
Conclusions:
Our findings provide the first evidence for adrenocortical tumors that CTCs may represent a useful marker to support differential diagnosis between ACC and ACA. The correlation with some clinical parameters suggests a possible relevance of CTC analysis for prognosis and noninvasive monitoring of disease progression and drug response.
Thoracic surgery in the UK

No abstract available
A new device for rapid isolation by size and characterization of rare circulating tumor cells

Background: Circulating tumor cells (CTCs) likely derive from clones in the primary tumor, suggesting that they can be used for all biological tests applying to the primary cells. Materials and Methods: The ScreenCell® devices are single-use and low-cost innovative devices that use a filter to isolate and sort tumor cells by size. Results: The ScreenCell® Cyto device is able to isolate rare, fixed, tumor cells, with a high recovery rate. Cells are well preserved morphologically. Immunocytochemistry and FISH assays can be performed directly on the filter. The ScreenCell® CC device allows isolation of live cells able to grow in culture. High quality genetic materials can be obtained directly from tumor cells isolated on the ScreenCell® MB device filter. Conclusion: Due to their reduced size, versatility, and capacity to isolate CTCs within minutes, the ScreenCell® devices may be able to simplify and improve non-invasive access to tumor cells.
Rapid and Simple Isolation of Circulating Tumor Cells for Clinical and Research Applications Using ScreenCell

Circulating tumor cells (CTCs) are malignant cells shed by the primary tumor or metastases into the peripheral circulation. CTCs give rise to distant metastases that are usually the ultimate cause of cancer-related death. A significant proportion of patients with early stage cancer in whom no metastases are identifiable will ultimately relapse as a result of hematogenous spread of tumor cells that were undetected at initial diagnosis and treatment.
Rapid separation of mononuclear hodgkin from multinuclear reed-sternberg cells

We describe a method to isolate small mononucleated Hodgkin (H) cells from multinucleated Reed Sternberg (RS) cells of Hodgkin lymphoma using the ScreenCell filter device. This filtration-based approach lends itself to future clinical applications in that it enables the separation of H and RS cells from lymph node biopsies, bone marrow aspirates, pleural effusions, and blood, including the isolation of monoclonal Hodgkin precursor cells from the blood.
Liquid Biopsy in Rare Cancers: Lessons from Hemangiopericytoma

Hemangiopericytoma (HPT) is a rare mesenchymal tumor of fibroblastic type and for its rarity is poorly studied. The most common sites of metastatic disease in patients with intracranial HPT are the bone, liver, and lung, suggestive for an hematogenous dissemination; for this reason, we investigated, for the first time, the presence of circulating tumor cells (CTCs) in hemangiopericytoma patient by CellSearch® and SceenCell® devices. Peripheral blood samples were drawn and processed by CellSearch, an EpCAM-dependent device, and ScreenCell®, a device size based. We found nontypical CTCs by CellSearch system and the immunofluorescence analysis performed on CTCs isolate by ScreenCell demonstrated the presence of single CTCs and CTC clusters. The molecular characterization of single CTCs and CTC clusters, using antibodies directed against EpCAM, CD34, cytokeratins (8, 18, and 19), and CD45, showed a great heterogeneity in CTC clusters. We believe that the present study may open a new scenario in the rare tumors: the introduction of the liquid biopsy and the molecular characterization of circulating tumor cells could lead to personalized targeted treatments and also for rare tumors.
Non-Metastatic Esophageal Adenocarcinoma: Circulating Tumor Cells in the Course of Multimodal Tumor Treatment

Background: Isolation of circulating tumor cells (CTC) holds the promise to improve response-prediction and personalization of cancer treatment. In this study, we test a filtration device for CTC isolation in patients with non-metastatic esophageal adenocarcinoma (EAC) within recent multimodal treatment protocols.
Methods: Peripheral blood specimens were drawn from EAC patients before and after neoadjuvant chemotherapy (FLOT)/chemoradiation (CROSS) as well as after surgery. Filtration using ScreenCell® devices captured CTC for cytologic analysis. Giemsa-stained specimens were evaluated by a cytopathologist; the cut-off was 1 CTC/specimen (6 mL). Immunohistochemistry with epithelial (pan-CK) and mesenchymal markers (vimentin) was performed.
Results: Morphologically diverse malignant CTCs were found in 12/20 patients in at least one blood specimen. CTCs were positive for both vimentin and pan-CK. More patients were CTC positive after neoadjuvant therapy (6/20 vs. 9/15) and CTCs per/ml increased in most of the CTC-positive patients. After surgery, 8/13 patients with available blood specimens were still CTC positive. In clinical follow-up, 5/9 patients who died were CTC-positive.
Conclusions: Detection of CTC by filtration within multimodal treatment protocols of non-metastatic EAC is feasible. The rate of CTC positive findings and the quantity of CTCs changes in the course of multimodal neoadjuvant chemoradiation/chemotherapy and surgery.
Cytopathological Heterogeneity of Circulating Tumor Cells in Non-metastatic Esophageal Adenocarcinoma

Background/aim: The presence of circulating tumor cells (CTC) has been reported to have an impact on prognosis in different tumor entities. Little is known about CTC morphology and heterogeneity.
Patients and methods: In a multicenter setting, pre-therapeutic peripheral blood specimens were drawn from patients with non-metastatic esophageal adenocarcinoma (EAC). CTCs were captured by size-based filtration (ScreenCell®), subsequently Giemsa-stained and evaluated by two trained readers. The isolated cells were categorized in groups based on morphologic criteria.
Results: Small and large single CTCs, as well as CTC-clusters, were observed in 69.2% (n=81) of the 117 specimens; small CTCs were observed most frequently (59%; n=69), followed by large CTCs (40%; n=47) and circulating cancer-associated macrophage-like cells (CAMLs; 34.2%, n=40). Clusters were rather rare (12%; n=14). CTC/CAML were heterogeneous in the cohort, but also within one specimen. Neither the presence of the CTC subtypes/CAMLs nor the exact cell count were associated with the primary clinical TNM stage.
Conclusion: Morphologically heterogenic CTCs and CAMLs are present in patients with non-metastatic, non-pretreated EAC.
Isolation and characterization of circulating melanoma cells by size filtration and fluorescent in-situ hybridization

Isolation of circulating tumor cells (CTCs) from blood of melanoma patients has been difficult owing to inconsistent expression of surface antigens. Here we report on the isolation, detection, and characterization of CTCs from blood of melanoma patients using microfiltration and fluorescent in-situ hybridization (FISH). Two tubes of blood from 15 patients with advanced melanoma were collected. These two tubes subsequently underwent filtration through a membrane with pore sizes of 7.5 μm. Isolated cells from one tube were analyzed by FISH for RREB1 (6p24), MYB (6q32), SE6 (D6Z1), and CCND1 (11q13) and the other paired specimen was analyzed by immunofluorescence for HMB45, melanoma-associated antigen recognized by T cells-1, tyrosinase and melanogenesis associated transcription factor. We identified CTCs in 10 out of 13 melanoma samples by immunofluorescence (2.5-99 CTCs/3 ml of blood) and in 13 specimens by FISH (7.2-76 CTCs/3 ml of blood) with more CTCs identified by FISH in 10 out 13 samples. Two filters failed. Our results show that CTCs are detectable in the majority of patients with advanced melanoma. These tools will be useful in characterizing treatment related changes of melanoma in CTCs.