We are transforming circulating tumor cell research into concrete solutions to improve cancer patient care.
Explore the full range of ScreenCell® CTC isolation kits, each specifically designed to meet the diverse needs of liquid biopsy applications.
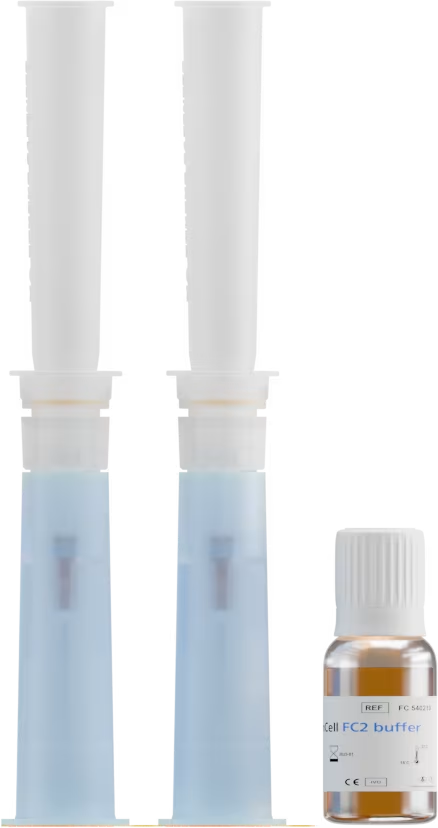
CYTO
Blood sample
< 4 hours
Cells fixed and analyzed on the isolation support

CSB
Blood sample
< 3 days
Cells fixed and analyzed on the isolation support
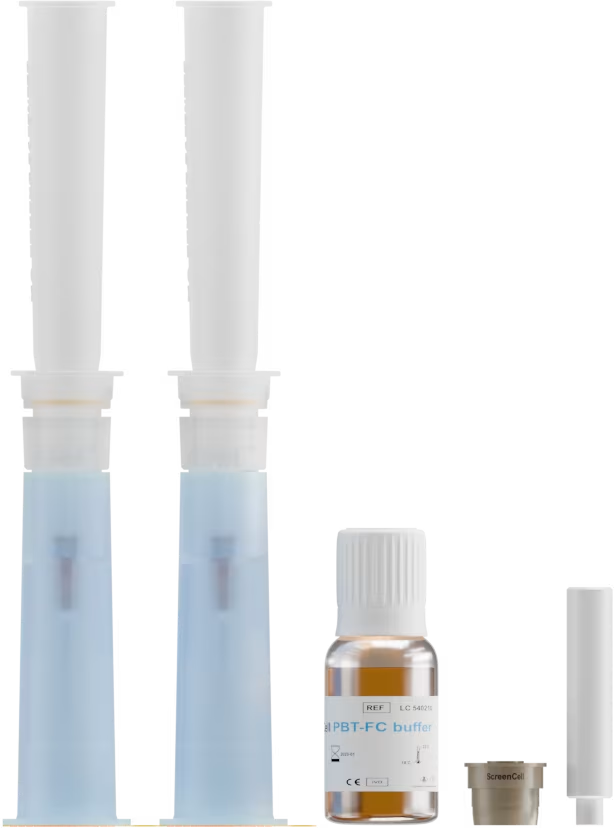
FCD
Blood sample
< 3 days
or
Cells fixed detached
from the isolation support for analysis in suspension or on slide
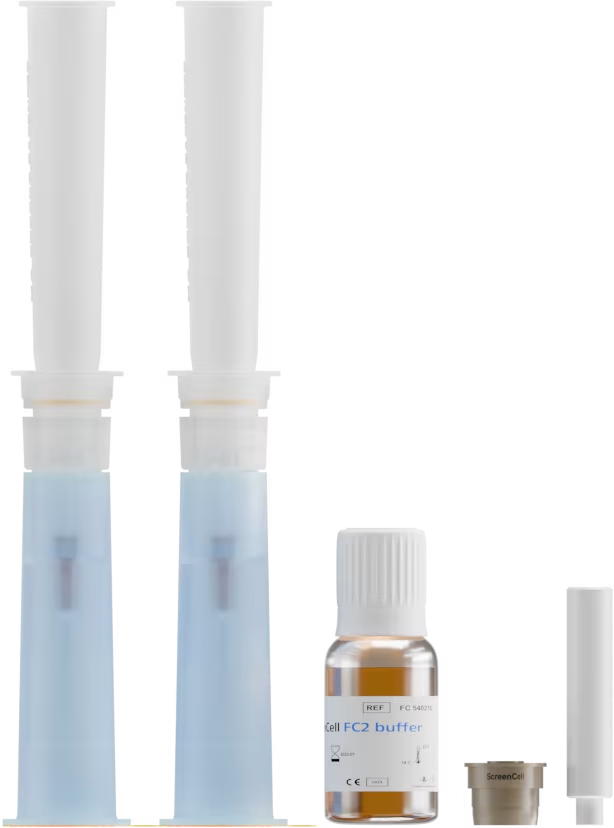
FCD-SP
Blood sample
< 4 hours
or
Cells fixed detached
from the isolation support for analysis in suspension or on slide

Blood sample
< 4 hours
Living cells lysed on the isolation support

Blood sample
< 3 days
Living cells lysed on the isolation support

Blood sample
< 4 hours
Living cells detached
from the isolation support for suspension analysis

Blood sample
< 4 hours
Living cells lysed on the isolation support

Blood sample
< 4 hours
Living cells detached
from the isolation support for suspension analysis
Fill in the fields below and we’ll take care of the rest.
Follow ScreenCell on LinkedIn